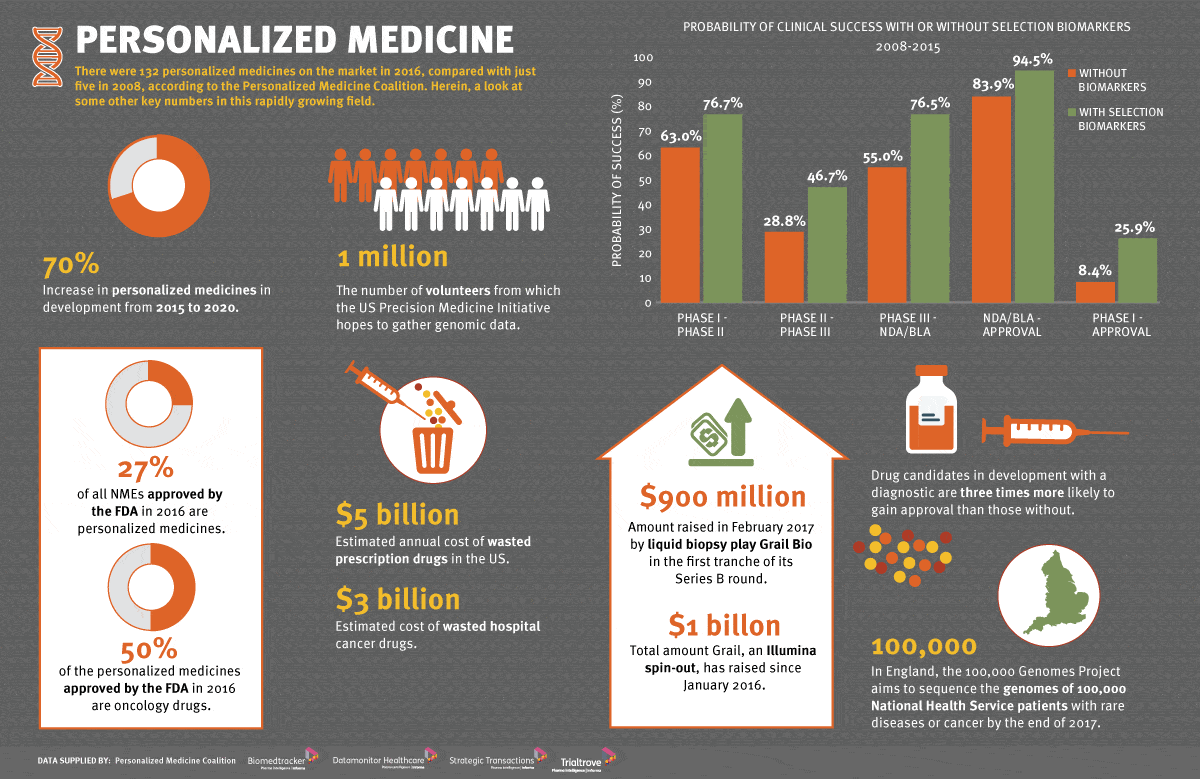
Foundation Medicine on X: "Now FDA approved as a companion diagnostic for PIQRAY® (alpelisib) to identify PIK3CA mutations, FoundationOne®CDx* may help match even more patients to approved treatments. Learn more: https://t.co/8lbnBl0RPX *Rx
FoundationOne®CDx Technical Information Foundation Medicine, Inc. 150 Second Street, Cambridge, MA 02141 Phone: 617.418.2200 I

Matthew Nelson on LinkedIn: Great achievement by the Foundation Medicine! Check out the latest FDA CDx…

Foundation Medicine and Pfizer Announce Broad Partnership to Develop Companion Diagnostics for Pfizer's Oncology Portfolio | Business Wire

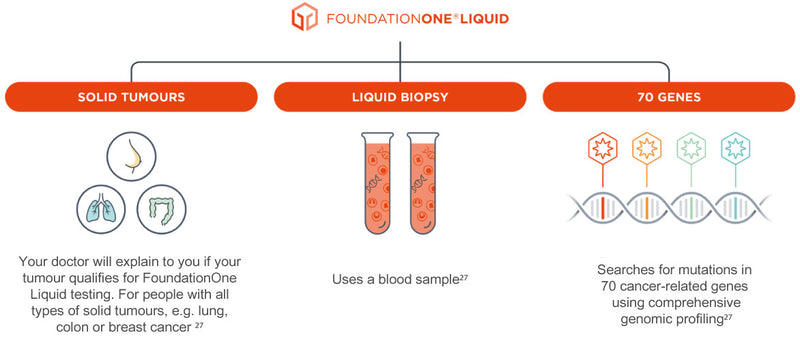
Roche Ireland on X: "FoundationOne®CDx uses a sample of your cancer tissue and FoundationOne®Liquid CDx a blood sample to provide a more complete picture of your cancer and help open up treatment

Comprehensive genomic profiling in oncology – from vision to reality | Latest news for Doctors, Nurses and Pharmacists | Pharmacy